What are ions?
Ions are atoms that gains or loses an electrons.
Ions are atoms that gains or loses an electrons.
How do ions bond?
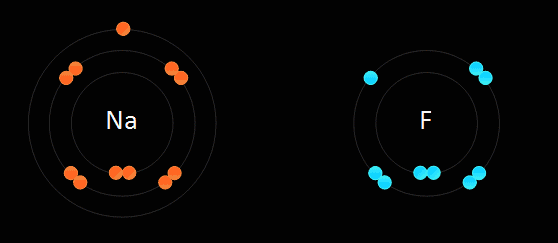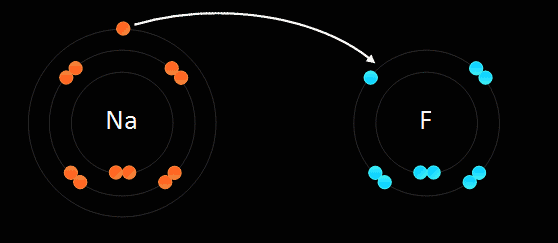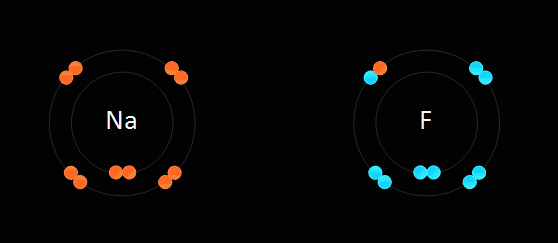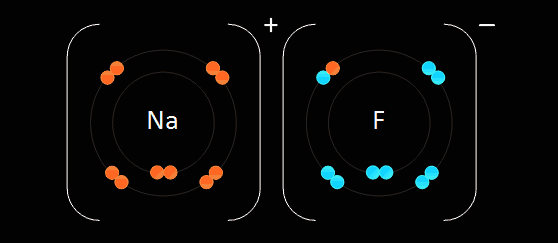
Ions bond when a cation, ions that have an overall positive charge, loses an electron to anions, ions that have an overall negative charge.
Electrostatic Force is the force that bonds cations and anions together, thus creating an ionic bond.
Ions bond when a cation, ions that have an overall positive charge, loses an electron to anions, ions that have an overall negative charge.
Electrostatic Force is the force that bonds cations and anions together, thus creating an ionic bond.
Why do ions bond?
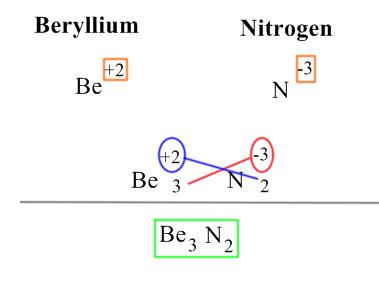
To become balanced. An ion can become balanced by either reaching a full valence level (8 electrons in its valence level ) or by obtaining an even number of electrons. The rules of this bonding are as followed:
Rules for bonding
Step 1. Identify an atom's oxidation number. Orange represents the oxidation number.
Step 2. The oxidation number should match between the two. If this is true, then proceed to Step 4.
Step 3. If the atom's oxidation numbers don't cancel each other out, then you must "cross" each atom's oxidation number, this can be seen in the picture below by the crossing of the Red and Blue.
Step 4. Congratulations, you have successfully balanced and bonded atoms together, this forms an ionic compound represented as Green in the picture below.
To become balanced. An ion can become balanced by either reaching a full valence level (8 electrons in its valence level ) or by obtaining an even number of electrons. The rules of this bonding are as followed:
Rules for bonding
Step 1. Identify an atom's oxidation number. Orange represents the oxidation number.
Step 2. The oxidation number should match between the two. If this is true, then proceed to Step 4.
Step 3. If the atom's oxidation numbers don't cancel each other out, then you must "cross" each atom's oxidation number, this can be seen in the picture below by the crossing of the Red and Blue.
Step 4. Congratulations, you have successfully balanced and bonded atoms together, this forms an ionic compound represented as Green in the picture below.
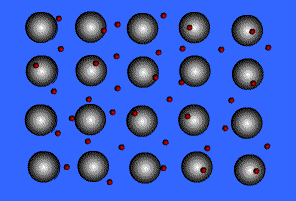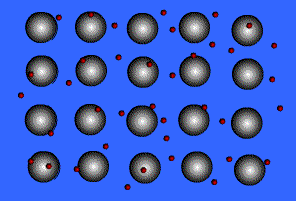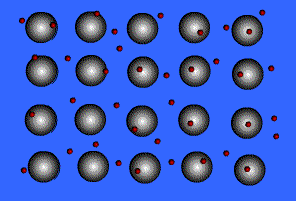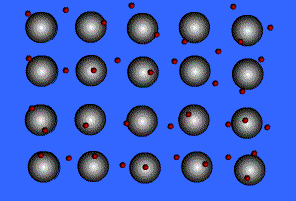
Metallic Bonding
Metals bond to other metals by sharing electrons, this creates an electron "sea" where an electron is constantly passed from one ion to the next. These shared electrons are known as delocalized electrons.
Metals bond to other metals by sharing electrons, this creates an electron "sea" where an electron is constantly passed from one ion to the next. These shared electrons are known as delocalized electrons.
Sources:
http://www.chemicool.com/the-periodic-table.html
http://www.docsity.com/en/news/education-2/types-chemical-bonding-presented-gifs/